Form 6-K - Report of foreign issuer [Rules 13a-16 and 15d-16]
January 06 2025 - 7:30AM
Edgar (US Regulatory)
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16 UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of January 2025
Commission File Number: 001-39032
PROFOUND MEDICAL CORP.
(Translation of registrant's name into English)
2400 Skymark Avenue, Unit 6, Mississauga, Ontario L4W 5K5
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F [ ] Form 40-F [ X ]
EXHIBIT INDEX
The following document is attached as an exhibit hereto and is incorporated by reference herein:
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | PROFOUND MEDICAL CORP. |
| | | (Registrant) |
| | | |
| | | |
| Date: January 6, 2025 | | /s/ Rashed Dewan |
| | | Rashed Dewan |
| | | Chief Financial Officer |
| | | |
EXHIBIT 99.1
Profound Medical to Present at the 43rd Annual J.P. Morgan Healthcare Conference
TORONTO, Jan. 06, 2025 (GLOBE NEWSWIRE) -- Profound Medical Corp. (NASDAQ:PROF; TSX:PRN) (“Profound” or the “Company”), a commercial-stage medical device company that develops and markets customizable, incision-free therapies for the ablation of diseased tissue, today announced that management will present an update on the Company’s business at the 43rd Annual J.P. Morgan Healthcare Conference on Thursday, January 16, 2025 at 11:15 a.m. Pacific Time in San Francisco, CA.
The presentation will be broadcast live and archived on the Company's website at www.profoundmedical.com under "Webcasts" in the Investors section.
About Profound Medical Corp.
Profound is a commercial-stage medical device company that develops and markets customizable, incision-free therapies for the ablation of diseased tissue.
Profound is commercializing TULSA-PRO®, a technology that combines real-time MRI, robotically-driven transurethral ultrasound and closed-loop temperature feedback control. The TULSA procedure, performed using the TULSA-PRO® system, has the potential of becoming a mainstream treatment modality across the entire prostate disease spectrum; ranging from low-, intermediate-, or high-risk prostate cancer; to hybrid patients suffering from both prostate cancer and benign prostatic hyperplasia (“BPH”); to men with BPH only; and also, to patients requiring salvage therapy for radio-recurrent localized prostate cancer. TULSA employs real-time MR guidance for precision to preserve patients’ urinary continence and sexual function, while killing the targeted prostate tissue via precise sound absorption technology that gently heats it to 55-57°C. TULSA is an incision- and radiation-free “one-and-done” procedure performed in a single session that takes a few hours. Virtually all prostate shapes and sizes can be safely, effectively, and efficiently treated with TULSA. There is no bleeding associated with the procedure; no hospital stay is required; and most TULSA patients report quick recovery to their normal routine. TULSA-PRO® is CE marked, Health Canada approved, and 510(k) cleared by the U.S. Food and Drug Administration (“FDA”).
Profound is also commercializing Sonalleve®, an innovative therapeutic platform that is CE marked for the treatment of uterine fibroids and palliative pain treatment of bone metastases. Sonalleve® has also been approved by the China National Medical Products Administration for the non-invasive treatment of uterine fibroids and has FDA approval under a Humanitarian Device Exemption for the treatment of osteoid osteoma. The Company is in the early stages of exploring additional potential treatment markets for Sonalleve® where the technology has been shown to have clinical application, such as non-invasive ablation of abdominal cancers and hyperthermia for cancer therapy.
Forward-Looking Statements
This release includes forward-looking statements regarding Profound and its business which may include, but is not limited to, the expectations regarding the efficacy of Profound’s technology in the treatment of prostate cancer, BPH, uterine fibroids, palliative pain treatment and osteoid osteoma; and the success of Profound’s U.S. commercialization strategy and activities for TULSA-PRO®. Often, but not always, forward-looking statements can be identified by the use of words such as "plans", "is expected", "expects", "scheduled", "intends", "contemplates", "anticipates", "believes", "proposes" or variations (including negative variations) of such words and phrases, or state that certain actions, events or results "may", "could", "would", "might" or "will" be taken, occur or be achieved. Such statements are based on the current expectations of the management of Profound. The forward-looking events and circumstances discussed in this release, may not occur by certain specified dates or at all and could differ materially as a result of known and unknown risk factors and uncertainties affecting the Company, including risks regarding the medical device industry, regulatory approvals, reimbursement, economic factors, the equity markets generally and risks associated with growth and competition. Although Profound has attempted to identify important factors that could cause actual actions, events or results to differ materially from those described in forward-looking statements, there may be other factors that cause actions, events or results to differ from those anticipated, estimated or intended. No forward-looking statement can be guaranteed. Except as required by applicable securities laws, forward-looking statements speak only as of the date on which they are made and Profound undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, or otherwise, other than as required by law.
For further information, please contact:
Stephen Kilmer
Investor Relations
skilmer@profoundmedical.com
T: 647.872.4849
Susan Thomas
Public Relations
sthomas@profoundmedical.com
T: 619.540.9195
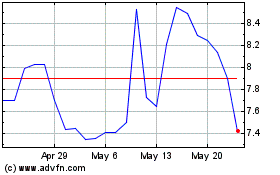
Profound Medical (NASDAQ:PROF)
Historical Stock Chart
From Dec 2024 to Jan 2025
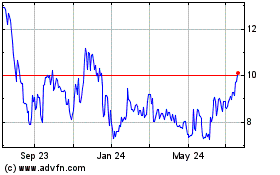
Profound Medical (NASDAQ:PROF)
Historical Stock Chart
From Jan 2024 to Jan 2025