UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE
ISSUER
PURSUANT TO RULE 13a-16
OR 15d-16 OF THE
SECURITIES EXCHANGE ACT
OF 1934
For the month of January 2024
Commission File Number: 001-39446
CureVac N.V.
(Exact Name of Registrant as Specified in Its
Charter)
Friedrich-Miescher-Strasse 15, 72076
Tübingen, Germany
+49 7071 9883 0
(Address of principal executive office)
Indicate by check mark whether the registrant files
or will file annual reports under cover of Form 20-F or Form 40-F:
On January 5, 2024, CureVac N.V. (the “Company”) issued
a press release announcing positive Phase 2 interim data from its COVID-19 vaccine development program in collaboration with GSK.
The information included in this Form 6-K
(including Exhibits 99.1 and 99.2, but excluding the statements of the Company’s Chief Development Officer contained in
Exhibit 99.1 hereto) is hereby incorporated by reference into the Company’s Registration Statement on Form F-3 (File No.
333-259613).
SIGNATURES
Pursuant to the requirements of the Securities
Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| |
CUREVAC N.V. |
| |
|
|
| |
By: |
/s/ Alexander
Zehnder |
| |
|
Chief Executive Officer |
Date: January 5, 2024
EXHIBIT INDEX
Exhibit 99.1

CureVac Announces
Positive Phase 2 Interim Data from COVID-19 Vaccine Development
Program in
Collaboration with GSK Providing Strong Validation
of Proprietary
Technology Platform
| • | Head-to-head
comparison with licensed bivalent mRNA-based comparator vaccine confirms competitive immune
responses at lower doses and favorable tolerability profile |
| • | Monovalent
mRNA vaccine candidate, CV0601, encoding Omicron BA.4-5 variant and bivalent candidate, CV0701,
encoding Omicron BA.4-5 variant as well as the original SARS-CoV-2 virus, successfully boosted
antibody titers and were generally well tolerated across all tested dose levels |
TÜBINGEN, Germany/BOSTON,
USA – January 5, 2024 – CureVac N.V. (Nasdaq: CVAC) (“CureVac”), a global biopharmaceutical company developing
a new class of transformative medicines based on messenger ribonucleic acid (“mRNA”), today announced positive interim data
from the ongoing Phase 2 study assessing monovalent and bivalent modified vaccine candidates against COVID-19. Both vaccine candidates
are being developed in collaboration with GSK. Selected data can be reviewed in the presentation associated with this press release.
Results from the formal
interim analysis showed that both vaccine candidates using CureVac’s proprietary second-generation mRNA backbone produced meaningful
immune responses and favorable reactogenicity profiles across all tested doses, including the lowest tested dose. All three of the dose
levels tested were below those used in mRNA-based COVID-19 vaccines licensed in the U.S. and EU.
“These positive Phase
2 data continue to strongly validate the competitiveness of our proprietary mRNA-technology platform and second-generation mRNA backbone
in comparison to a licensed mRNA-based vaccine,” said Dr. Myriam Mendila, Chief Development Officer of CureVac. “We are greatly
encouraged by the strong immunogenicity results achieved for our COVID-19 mRNA vaccine candidates and are in advanced discussions with
regulatory authorities to determine the best path forward for a pivotal Phase 3 study. With this, we advance our joint COVID-19 development
program along with our joint flu vaccine program, which continues to progress steadily as well.”
The Phase 2 study assesses
the safety and immunogenicity of different single booster doses of monovalent vaccine candidate CV0601, encoding the spike protein of
the Omicron BA.4-5 variant and bivalent vaccine candidate CV0701, encoding the spike protein of the Omicron BA.45 variant and original
SARS-CoV-2 virus. Safety and immunogenicity were assessed in comparison to a licensed bivalent mRNA-based COVID-19 comparator vaccine.
While the monovalent candidate CV0601 was tested at a single medium dose level, the bivalent candidate CV0701 was tested at low, medium,
and high dose levels. The study is being conducted in Australia and is fully enrolled with 427 healthy adults aged 18 and older equally
randomized between dose groups.

Reactogenicity data cover
all dose groups for both vaccine candidates. The vaccine candidates were shown to be generally well tolerated with a lower or similar
proportion of participants reporting solicited adverse events when compared to comparator vaccine participants within seven days of dosing.
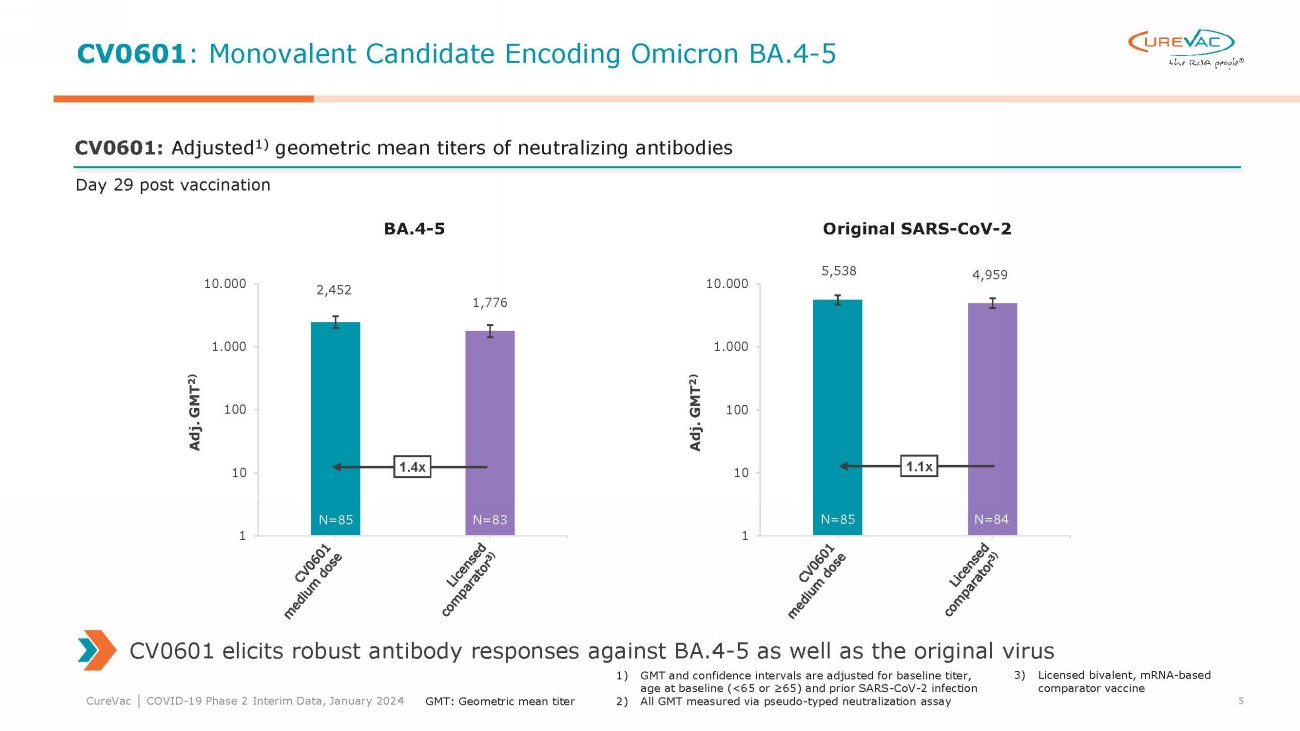
Interim immunogenicity data
showed meaningful titers of neutralizing antibodies for both candidates at all dose levels. Titers of neutralizing antibodies matched
or numerically exceeded the titers induced by the licensed comparator vaccine at all tested doses except for the low dose level of CV0701.
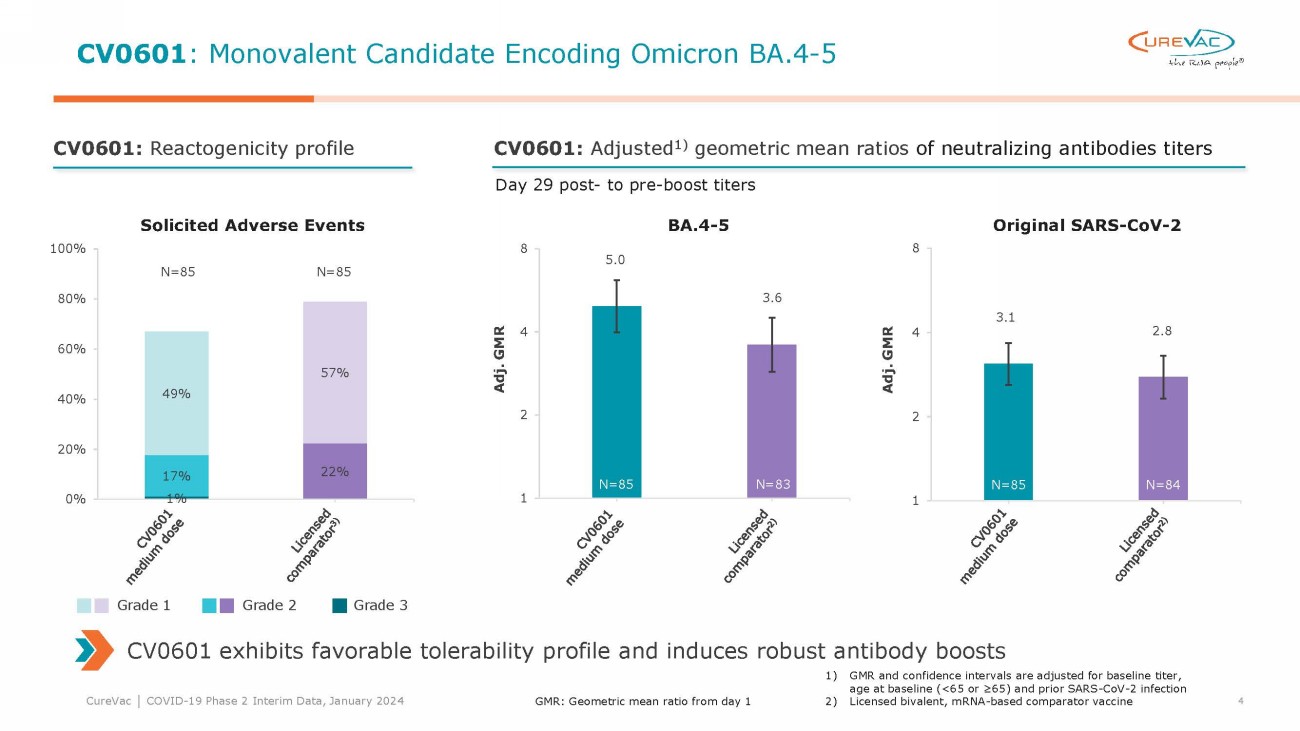
The monovalent candidate
CV0601, which was tested at a medium dose level, elicited neutralizing antibody titers against the Omicron BA.4-5 variant on day 29 following
the booster vaccination that were 5.0 times the pre-boosting titers, numerically exceeding the 3.6-fold ratio generated by the licensed
comparator vaccine.
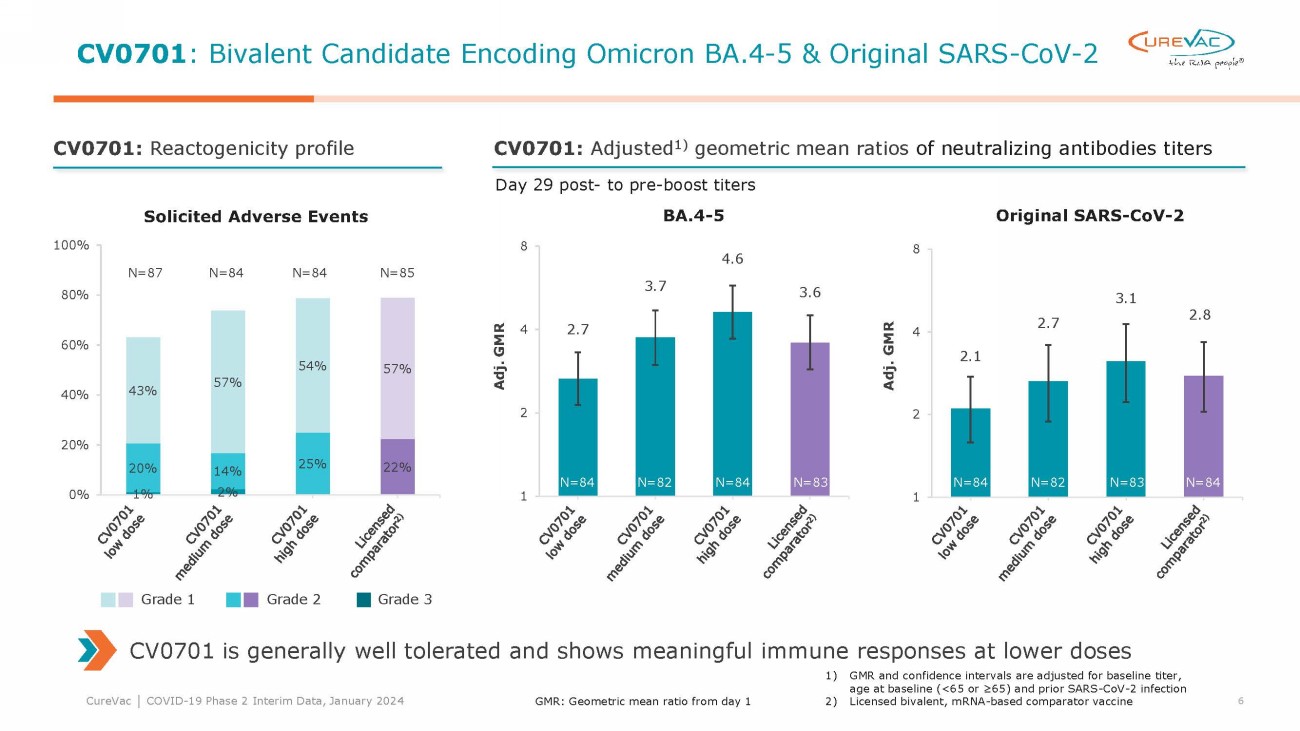
For the low, medium, and
high dose levels tested for the bivalent candidate CV0701, neutralizing antibody titers against BA.4-5 on day 29 following the booster
vaccination were 2.7-fold, 3.7-fold and 4.6-fold the titers before the booster, compared to a 3.6-fold ratio of post- to prebooster titers
for the comparator vaccine.
About CureVac
CureVac (Nasdaq: CVAC) is
a global biopharmaceutical company in the field of messenger RNA (mRNA) technology, with more than 20 years of expertise in developing,
optimizing, and manufacturing this versatile biological molecule for medical purposes. The principle of CureVac’s proprietary technology
is the use of optimized mRNA as a data carrier to instruct the human body to produce its own proteins capable of fighting a broad range
of diseases. In July 2020, CureVac entered in a collaboration with GSK to jointly develop new products in prophylactic vaccines for infectious
diseases based on CureVac’s second-generation mRNA technology. This collaboration was later extended to the development of second-generation
COVID-19 vaccine candidates, and modified mRNA vaccine technologies. Based on its proprietary technology, CureVac has built a deep clinical
pipeline across the areas of prophylactic vaccines, cancer therapies, antibody therapies, and the treatment of rare diseases. CureVac
N.V. has its headquarters in Tübingen, Germany, and has more than 1,100 employees across its sites in Germany, the Netherlands,
Belgium, Switzerland and the U.S. Further information can be found at www.curevac.com.
CureVac Media and Investor
Relations Contact
Dr. Sarah Fakih, Vice President
Corporate Communications and Investor Relations
CureVac, Tübingen,
Germany
T: +49 7071 9883-1298
M: +49 160 90 496949
sarah.fakih@curevac.com

Forward-Looking Statements CureVac
This press release contains
statements that constitute “forward looking statements” as that term is defined in the United States Private Securities Litigation
Reform Act of 1995, including statements that express the opinions, expectations, beliefs, plans, objectives, assumptions or projections
of CureVac N.V. and/or its wholly owned subsidiaries CureVac SE, CureVac Manufacturing GmbH, CureVac Inc., CureVac Swiss AG, CureVac
Corporate Services GmbH, CureVac RNA Printer GmbH, CureVac Belgium SA and CureVac Netherlands B.V. (the “company”) regarding
future events or future results, in contrast with statements that reflect historical facts. Examples include discussion of the potential
efficacy of the company’s vaccine and treatment candidates and the company’s strategies, financing plans, growth opportunities
and market growth. In some cases, you can identify such forward-looking statements by terminology such as “anticipate,” “intend,”
“believe,” “estimate,” “plan,” “seek,” “project,” or “expect,”
“may,” “will,” “would,” “could,” “potential,” “intend,” or “should,”
the negative of these terms or similar expressions. Forward-looking statements are based on management’s current beliefs and assumptions
and on information currently available to the company. However, these forward-looking statements are not a guarantee of the company’s
performance, and you should not place undue reliance on such statements. Forward-looking statements are subject to many risks, uncertainties
and other variable circumstances, including negative worldwide economic conditions and ongoing instability and volatility in the worldwide
financial markets, ability to obtain funding, ability to conduct current and future preclinical studies and clinical trials, the timing,
expense and uncertainty of regulatory approval, reliance on third parties and collaboration partners, ability to commercialize products,
ability to manufacture any products, possible changes in current and proposed legislation, regulations and governmental policies, pressures
from increasing competition and consolidation in the company’s industry, the effects of the COVID-19 pandemic on the company’s
business and results of operations, ability to manage growth, reliance on key personnel, reliance on intellectual property protection,
ability to provide for patient safety, fluctuations of operating results due to the effect of exchange rates, delays in litigation proceedings,
different judicial outcomes or other factors. Such risks and uncertainties may cause the statements to be inaccurate and readers are
cautioned not to place undue reliance on such statements. Many of these risks are outside of the company’s control and could cause
its actual results to differ materially from those it thought would occur. The forward-looking statements included in this press release
are made only as of the date hereof. The company does not undertake, and specifically declines, any obligation to update any such statements
or to publicly announce the results of any revisions to any such statements to reflect future events or developments, except as required
by law.
For further information,
please reference the company’s reports and documents filed with the U.S. Securities and Exchange Commission (SEC). You may get
these documents by visiting EDGAR on the SEC website at www.sec.gov.
Exhibit 99.2




CureVac NV (NASDAQ:CVAC)
Historical Stock Chart
From Dec 2024 to Jan 2025
CureVac NV (NASDAQ:CVAC)
Historical Stock Chart
From Jan 2024 to Jan 2025